- Home
- About Us
- Work
- Journal
- Contact
- Audacity mac core audio input choices
- The binding of isaac online multiplayert
- Binding of isaac afterbirth mods osx
- Free rar to zip converter download
- Google blackberry desktop manager
- Accessing alt key on mac
- Watch kung fu panda 1 online free
- Hollywood hindi dubbed movies download in hd for pc
- Swift shader 3-0 rar download 64 bit
- Arduino i2c example program
- Ati radeon hd 3450 driver windows 10 dual monitor support
- Instagram bulk downloader extension
- External blu ray burner pc world
- Evernote premium mac
- X2x free mp3 converter
- Best screen recorder and editor for pc
- Screenplays wanted by producers 2017
- Gembox chart examples
- Connect macbook to monitor wirelessly
- Tamil movies download 2020 isaimini
- Vmware fusion 8 pro download
- Zoho outlook settings pop
- Joe budden hits drake girlfriend
- Best picture stacking software for astrophotography
- Advanced ip scanner portable
- 800 mmhg to atm
- Home
- About Us
- Work
- Journal
- Contact
- Audacity mac core audio input choices
- The binding of isaac online multiplayert
- Binding of isaac afterbirth mods osx
- Free rar to zip converter download
- Google blackberry desktop manager
- Accessing alt key on mac
- Watch kung fu panda 1 online free
- Hollywood hindi dubbed movies download in hd for pc
- Swift shader 3-0 rar download 64 bit
- Arduino i2c example program
- Ati radeon hd 3450 driver windows 10 dual monitor support
- Instagram bulk downloader extension
- External blu ray burner pc world
- Evernote premium mac
- X2x free mp3 converter
- Best screen recorder and editor for pc
- Screenplays wanted by producers 2017
- Gembox chart examples
- Connect macbook to monitor wirelessly
- Tamil movies download 2020 isaimini
- Vmware fusion 8 pro download
- Zoho outlook settings pop
- Joe budden hits drake girlfriend
- Best picture stacking software for astrophotography
- Advanced ip scanner portable
- 800 mmhg to atm

Symbols, abbreviations, or full names for units of length,Īrea, mass, pressure, and other types. You have been called to the home of a 62-year-old male who has failed to go to dialysis for the past week and is now unresponsive. You can find metric conversion tables for SI units, as wellĪs English units, currency, and other data. It is approximately equal to Earth's atmospheric pressure at sea level.Ĭonversion calculator for all types of measurement units. It is sometimes used as a reference pressure or standard pressure. color(white)(aaaaaaaaaaaaaaaaaaaa)color(blue)'1 atm'/'760 mm Hg' Knowing what our conversion factor is, we can now solve by setting up the problem as so, ('0.800' cancel'atm') /'1'('760 mm Hg')/('1' cancel'atm') 'answer' which is simply plugging in our knowns and getting out our answer in 'mm Hg'. The standard atmosphere (symbol: atm) is a unit of pressure defined as 101325 Pa (1.01325 bar). The pressure p in atmospheres (atm) is equal to the pressure p in millimeter mercury (0c) (mmHg) times 0.
800 MMHG TO ATM HOW TO
How to convert Millimeter Mercury (0C) to Atmospheres 1 millimeter mercury (mmHg) is equal to 0.00132 atmospheres (atm). The unit is named after Evangelista Torricelli, Italian physicist and mathematician, for his discovery of the principle of the barometer in 1643. 800 Millimeter Mercury (0C) 1.05263 Atmospheres. at what pressure will 0.25 mol of o2 occupy a volume of 2.0 l at 80 c. torr The average pressure of an inhaler is 400 kPa. a 45 ml sample of a gas at 10 and 720 torr weighs 0.051 g what is the molar mass of the gas. Convert the pressure of a standard car tire (32.0 psi) to torr. if the density of a gas is 1.2g/ml at 745 torr. calculate the density of ammonia gas at 27 c and 635 torr. atmlbf/ft2 .220402 lbf/ft2 atmlbf/yd2 5.983614 lbf/yd2 atmtorr 1 atm 760 torr atmcmHg 1 atm 76 cmHg atmmmHg 1 atm 760 mmHg atminHg 1 atm 29.921373 inHg atmInch mercury (60F) coefficient: 30.005777 atminAg 1 atm 406.793724 inAg atmftAg 1 atm 33. It is the atmospheric pressure that supports a column of mercury 1 millimetre high. calculate the density of nh3 at 850 torr and 100.
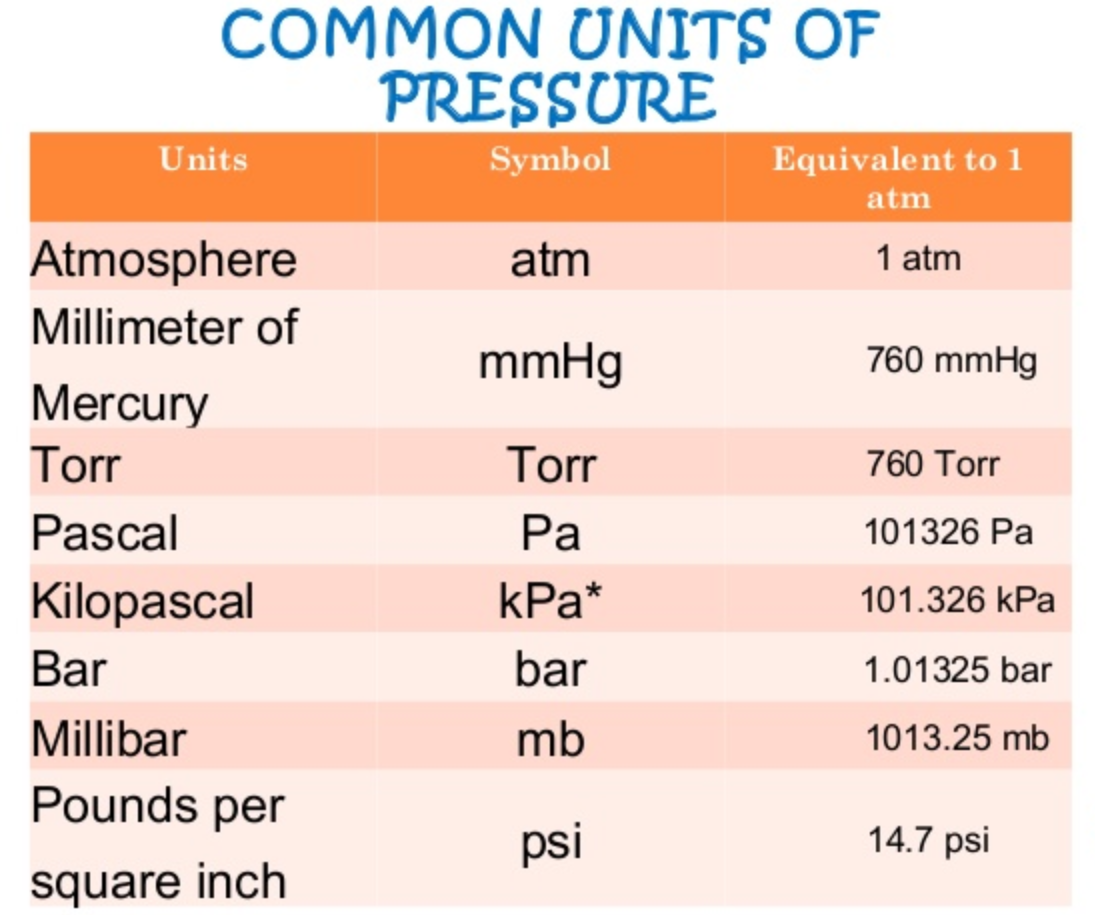
The torr (symbol: Torr) or millimetre of mercury (mmHg) is a non-SI unit of pressure. If the final temperature is 30.° C, the final volume is 5.7 L, and the final pressure is 800. The atmospheric pressure at sea level is 14.7 psi.You can do the reverse unit conversion fromĪtm to mmHg, or enter any two units below: Enter two units to convert From: A sample of argon has a volume of 5.0 dm3 and the pressure is 0.92 atm. Atmospheric pressure is also often stated as pounds/square inch (psi). Standard atmospheric pressure is called 1 atm of pressure and is equal to 760 mmHg and 101.3 kPa.
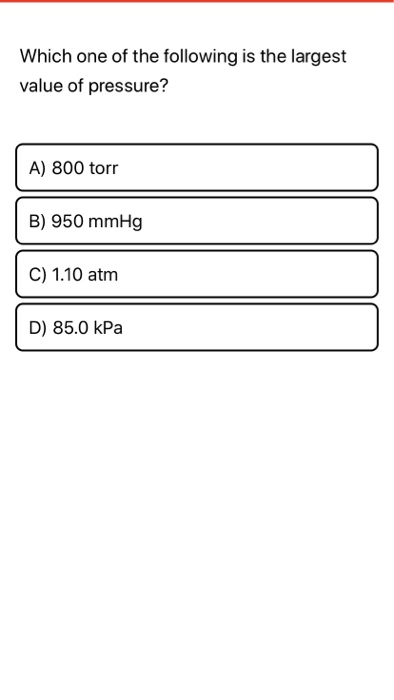

Another commonly used unit of pressure is the atmosphere (atm). A pascal is a very small amount of pressure, so the more useful unit for everyday gas pressures is the kilopascal (kPa). The pascal (Pa) is the standard unit of pressure. Both torr and mmHg are units of measurement of pressure. An equivalent unit to the mmHg is called the torr, in honor of the inventor of the barometer, Evangelista Torricelli. The key difference between torr and mmHg is that the value of one torr can be obtained by dividing 101325 from 760, whereas the value of one mmHg is given exactly as 133.3224. What volume would 8.00 g of oxygen gas (O2) gas occupy at 45.0☌ and a pressure of 2. One unit of gas pressure is the millimeter of mercury (mmHg). What is the Celsius temperature of 100.0 g of chlorine gas, Cl2, in a 40.0-L container at 800 mm Hg Calculate the volume occupied by 18.0 g of water vapor at 100 ☌ and 1.00 atm. Tire pressure is best measured when the tire is cold since driving the car for a while will heat up the air in the tire and increase the pressure.Ī barometer measures gas pressure by the height of the column of mercury. Here is a repeat from the 'Four Variables' file:There are three different units of pressure used in chemistry. The pressure on the tire is the maximum pressure for that tire, not the recommended one.
800 MMHG TO ATM MANUAL
The recommended pressure for that model of car (usually somewhere between 32-35 psi) is usually listed in the owner’s manual or stamped somewhere inside the door. NameDate Period Mixed Gas Law Review Worksheet 1) A 35 L sample of gas is initially at 15oC and 800.0 mmHg. Standard atmospheric pressure is called 1 atm of pressure and is equal to 760 mmHg and 101. The car gets better gas mileage and the tires don’t wear out as fast. View MixedGasLawPractice.docx from CHEMISTRY Chemistry at Northview High School. Another commonly used unit of pressure is the atmosphere (atm). Una señora reparte entre sus nietos en partes directamente proporcionalmente a sus eadades de sus tierras. Publicidad Publicidad Nuevas preguntas de Química. The ride is smoother and safer than with lowered pressure. convertir 800 mmhg a atm 1 atm - 760 mmHg x - 800 mmHg x 800 mmHg x 1 atm 760 mmHg x 1.052 atm. There are several benefits to maintaining the proper air pressure in a car tire.
